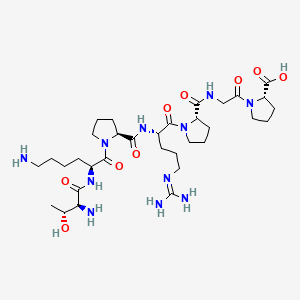
Selank
Selank (Thr-Lys-Pro-Arg-Pro-Gly-Pro)
Research Hub — Aggregated Studies
MedTech Research Group aggregates published research from peer-reviewed journals, clinical trials, and academic institutions. We do not conduct original research. All studies cited below are the work of their respective authors and institutions. Sources are linked for verification.
This product is designated FOR RESEARCH USE ONLY (RUO). These compounds have not been approved or cleared under 21 U.S.C. § 505 and have not been evaluated by the FDA for safety, efficacy, or labeling for clinical, diagnostic, or therapeutic use in humans or animals.
MedTech Research Group will only fulfill orders to qualified researchers affiliated with accredited academic institutions, licensed research facilities, or organizations with active IRB/IACUC oversight.
Purchaser Restrictions
- Purchaser must be a qualified researcher at an accredited institution or licensed research facility
- This product may not be sold or redistributed to individual consumers, wellness clinics, health food stores, or retail establishments
- Not intended for human or animal consumption, diagnostic use, or therapeutic application
- Institutional affiliation and research purpose will be verified prior to order fulfillment
Distribution is limited to qualified research use in compliance with applicable federal and state law. These products bear the "For Research Use Only" designation per FDA labeling requirements (minimum 10 pt. font). Ref: 21 U.S.C. § 505; FD&C Act § 201(p) (unapproved new drug definition).
| Risk Tier | GREEN |
| Category | Neuropeptides |
| Subcategory | Anxiolytic / Immunomodulatory |
| Pharmacological Class | Synthetic Peptide |
| Subclass | Tuftsin Analog with Pro-Gly-Pro Extension |
| Molecular Type | Synthetic Heptapeptide (7 amino acids: Thr-Lys-Pro-Arg-Pro-Gly-Pro) |
| Origin | Synthetic — designed at the Institute of Molecular Genetics, Russian Academy of Sciences; based on endogenous tuftsin (Thr-Lys-Pro-Arg) with C-terminal Pro-Gly-Pro stability extension |
| Regulatory Status | Approved in Russia as an anxiolytic and nootropic medication. Not approved in the US, EU, or other Western regulatory jurisdictions. |
| Route of Administration | Subcutaneous injection, intranasal (approved route in Russia) |
| Reconstitution | Lyophilized powder; reconstitute with bacteriostatic water |
| Storage | Refrigerate (2-8°C) |
Chemical Properties
| Molecular Formula | C33H57N11O9 |
| Molecular Weight | 751.9 g/mol |
| Exact Mass | 751.43407244 Da |
| InChI Key | JTDTXGMXNXBGBZ-YVHUGQOKSA-N |
| Synonyms |
|
| PubChem | View full record |
Source: NCBI PubChem — public domain data
2D structure diagram from NCBI PubChem. This is the actual molecular structure of Selank.
Description
Selank is a synthetic heptapeptide designed as a stabilized analog of tuftsin, an endogenous immunomodulatory tetrapeptide (Thr-Lys-Pro-Arg) that is naturally produced by the enzymatic cleavage of the Fc region of IgG antibodies in the spleen. Tuftsin was discovered in the 1970s and is known to stimulate phagocytosis, natural killer cell activity, and monocyte chemotaxis. However, native tuftsin has a very short half-life (minutes) due to rapid enzymatic degradation. Selank was created by appending a Pro-Gly-Pro tripeptide to the C-terminus of tuftsin — this glyproline extension is a common strategy in Russian peptide pharmacology (also used in Semax) that dramatically increases resistance to peptidase degradation and extends the functional half-life.
The anxiolytic mechanism of Selank is multifaceted and involves modulation of several neurotransmitter systems. It has been shown to: (1) modulate GABA-A receptor function, enhancing benzodiazepine-site binding and GABAergic neurotransmission in the brain (particularly in the amygdala and hippocampus); (2) influence serotonergic signaling by modulating the expression of 5-HT1A receptors and serotonin metabolism; (3) affect the expression of brain-derived neurotrophic factor (BDNF) and nerve growth factor (NGF) — both critical for neuronal survival and plasticity; (4) modulate the enkephalin/opioid peptide system; and (5) influence gene expression of approximately 36 genes related to neurotransmitter signaling, immune function, and stress response. Unlike benzodiazepines, Selank does not cause sedation, muscle relaxation, cognitive impairment, or dependence — it is described as producing a "clear-headed" anxiolysis with preserved or enhanced cognitive function.
Clinical Context
Selank is approved and prescribed in Russia as an anxiolytic medication, typically administered as a nasal spray (0.15% solution). Russian clinical studies report efficacy comparable to benzodiazepines for generalized anxiety disorder (GAD) but without the sedation, cognitive impairment, tolerance, or withdrawal syndrome associated with benzodiazepines. It is also reported to have nootropic (cognitive-enhancing) effects, including improved memory, attention, and information processing. The dual anxiolytic-immunomodulatory profile is unique and stems from its tuftsin-derived structure. Selank represents one of the most affordable peptides in the catalog ($37.04/10mg at $50 MSRP).
- Approved anxiolytic in Russia — extensive clinical use but limited Western clinical trial data
- Non-sedating, non-addictive, no withdrawal — fundamentally different from benzodiazepine anxiolysis
- Dual anxiolytic + immunomodulatory mechanism due to tuftsin backbone
- Intranasal route is the approved delivery method in Russia; subcutaneous injection also used in research
- Well-tolerated with minimal adverse effects reported
- Narrow MSRP margin ($37.04 cost vs. $50 MSRP) — low-margin product
- Not a benzodiazepine, not GABAergic in the traditional sense — modulates GABA-A allosterically rather than directly
- Can be combined with Semax for synergistic anxiolytic + nootropic effects (common Russian clinical practice)
Research data sourced from UniProt. CC BY 4.0 — attribution required.
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
Amino acid sequence length: 4 residues
Published Research
Published Research & Clinical Data
Peer-reviewed studies and clinical trial data related to Selank
20 from PubChem
All research below is conducted by independent institutions. MedTech Research Group provides these references for informational purposes only.
[Semax and selank inhibit the enkephalin-degrading enzymes from human serum]].
Kost NV, Sokolov OIu, Gabaeva MV, Grivennikov IA, Andreeva LA, et al.. Bioorganicheskaia khimiia, 2001.PMID: 11443939
Zozulya AA, Kost NV, Yu Sokolov O, Gabaeva MV, Grivennikov IA, et al.. Bulletin of experimental biology and medicine, 2001.PMID: 11550013
Kozlovskaia MM, Kozlovskiĭ II, Val'dman EA, Seredenin SB. Rossiiskii fiziologicheskii zhurnal imeni I.M. Sechenova, 2002.PMID: 12154572
Sokolov OY, Meshavkin VK, Kost NV, Zozulya AA. Bulletin of experimental biology and medicine, 2002.PMID: 12432865
[Optimizing action of synthetic peptide Selank on active avoidance conditioning test in rats].
Kozlovskiĭ II, Danchev ND. Zhurnal vysshei nervnoi deiatelnosti imeni I P Pavlova, 2002.PMID: 12449836
Kozlovskii II, Danchev ND. Neuroscience and behavioral physiology, 2003.PMID: 14552529
Selank and short peptides of the tuftsin family in the regulation of adaptive behavior in stress.
Kozlovskaya MM, Kozlovskii II, Val'dman EA, Seredenin SB. Neuroscience and behavioral physiology, 2003.PMID: 14969422
Zolotarev IuA, Sokolov OIu, Kost NV, Vas'kovskiĭ BV, Miasoedov NF, et al.. Bioorganicheskaia khimiia, 2004.PMID: 15344652
A new property of the synthetic anxiolytic Selank and its derivatives.
Pavlov TS, Samonina GE, Andreeva LA, Myasoedov NF, Ashmarin IP. Doklady biological sciences : proceedings of the Academy of Sciences of the USSR, Biological sciences sections, 2004.PMID: 15508574
Pavlov TS, Sanzhieva LTs, Samonina GE, Sergeev VI, Lelekova TV. Rossiiskii fiziologicheskii zhurnal imeni I.M. Sechenova, 2005.PMID: 15835541
Gan'shina TS, Kozlovskiĭ II. Eksperimental'naia i klinicheskaia farmakologiia, 2005.PMID: 16193654
Liapina LA, Pastorova VE, Obergan TIu, Samonina GE, Ashmarin IP, et al.. Izvestiia Akademii nauk. Seriia biologicheskaia, 2006.PMID: 16634437
[Evenly tritium-labeled peptides and their in vivo and in vitro biodegradation].
Zolotarev IuA, Dadaian AK, Dolotov OV, Kozik VS, Kost NV, et al.. Bioorganicheskaia khimiia, 2006.PMID: 16637290
Seasonal effects of Selank on the behavior of hibernating animals.
Semenova TP, Kozlovskaya MM, Zuikov AV, Kozlovskii II, Andreeva LA. Bulletin of experimental biology and medicine, 2005.PMID: 16848230
Semenova TP, Kozlovskaia MM, Zuĭkov AV, Kozlovskiĭ II, Zakharova NM, et al.. Rossiiskii fiziologicheskii zhurnal imeni I.M. Sechenova, 2006.PMID: 17385425
Meshavkin VK, Kost NV, Sokolov OY, Zolotarev YA, Myasoedov NF, et al.. Bulletin of experimental biology and medicine, 2006.PMID: 17415472
Selank and its metabolites maintain homeostasis in the gastric mucosa.
Pavlov TS, Samonina GE, Bakaeva ZV, Zolotarev YA, Guseva AA. Bulletin of experimental biology and medicine, 2007.PMID: 18019011
Semenova TP, Kozlovskaya MM, Zuikov AV, Kozlovskii II, Zakharova NM, et al.. Neuroscience and behavioral physiology, 2008.PMID: 18197389
Zozulia AA, Neznamov GG, Siuniakov TS, Kost NV, Gabaeva MV, et al.. Zhurnal nevrologii i psikhiatrii imeni S.S. Korsakova, 2008.PMID: 18454096
Kozlovskiĭ II, Belozertsev IuF, Semenova TP, Zuĭkov AV, Kozlovskaia MM. Eksperimental'naia i klinicheskaia farmakologiia, 2008.PMID: 18488898
Research Library — 175 Papers
Research data sourced from OpenAlex. CC0 public domain. Articles are the work of their respective authors.
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
Natural and hybrid (“chimeric”) stable regulatory glyproline peptides
И. П. Ашмарин, Г. Е. Самонина, L. A. Lyapina, et al. · Pathophysiology
Research by И. П. Ашмарин et al., published in Pathophysiology. Not conducted by MedTech Research Group.
Neuroprotective Peptides and New Strategies for Ischemic Stroke Drug Discoveries
Lyudmila V. Dergunova, Ivan B. Filippenkov, Limborskaia Sa, et al. · Genes
Research by Lyudmila V. Dergunova et al., published in Genes. Not conducted by MedTech Research Group.
A New Generation of Drugs: Synthetic Peptides Based on Natural Regulatory Peptides
Timur Kolomin, М. И. Шадрина, P. A. Slominsky, et al. · Neuroscience & Medicine
Research by Timur Kolomin et al., published in Neuroscience & Medicine. Not conducted by MedTech Research Group.
Sedative‐Hypnotic Agents That Impact Gamma‐Aminobutyric Acid Receptors: Focus on Flunitrazepam, Gamma‐Hydroxybutyric Acid, Phenibut, and Selank
Cassandra R. Doyno, C Michael White · The Journal of Clinical Pharmacology
Research by Cassandra R. Doyno et al., published in The Journal of Clinical Pharmacology. Not conducted by MedTech Research Group.
Role of BDNF in Neuroplasticity Associated with Alcohol Dependence
Danil Peregud, Valeria Yu. Baronets, Natalia N. Terebilina, et al. · Biochemistry (Moscow)
Research by Danil Peregud et al., published in Biochemistry (Moscow). Not conducted by MedTech Research Group.
Enhanced Mucosal Immune Responses Induced by a Combined Candidate Mucosal Vaccine Based on Hepatitis A Virus and Hepatitis E Virus Structural Proteins Linked to Tuftsin
Yan Gao, Qiudong Su, Yi Yao, et al. · PLoS ONE
Research by Yan Gao et al., published in PLoS ONE. Not conducted by MedTech Research Group.
Selank and Short Peptides of the Tuftsin Family in the Regulation of Adaptive Behavior in Stress
M. M. Kozlovskaya, И. И. Козловский, Е А Вальдман, et al. · Neuroscience and Behavioral Physiology
Research by M. M. Kozlovskaya et al., published in Neuroscience and Behavioral Physiology. Not conducted by MedTech Research Group.
Selank Administration Affects the Expression of Some Genes Involved in GABAergic Neurotransmission
Anastasiya Volkova, М. И. Шадрина, Timur Kolomin, et al. · Frontiers in Pharmacology
Research by Anastasiya Volkova et al., published in Frontiers in Pharmacology. Not conducted by MedTech Research Group.
Neuropsychopharmacological profiling of scoparone in mice
Joanna Kowalczyk, Barbara Budzyńska, Łukasz Kurach, et al. · Scientific Reports
Research by Joanna Kowalczyk et al., published in Scientific Reports. Not conducted by MedTech Research Group.
Effect of chronic unpredictable stress on mice with developmental under-expression of the Ahi1 gene: behavioral manifestations and neurobiological correlates
Gilly Wolf, Tzuri Lifschytz, Hagar Ben-Ari, et al. · Translational Psychiatry
Research by Gilly Wolf et al., published in Translational Psychiatry. Not conducted by MedTech Research Group.