Sermorelin
Research Hub — Aggregated Studies
MedTech Research Group aggregates published research from peer-reviewed journals, clinical trials, and academic institutions. We do not conduct original research. All studies cited below are the work of their respective authors and institutions. Sources are linked for verification.
This product is designated FOR RESEARCH USE ONLY (RUO). These compounds have not been approved or cleared under 21 U.S.C. § 505 and have not been evaluated by the FDA for safety, efficacy, or labeling for clinical, diagnostic, or therapeutic use in humans or animals.
MedTech Research Group will only fulfill orders to qualified researchers affiliated with accredited academic institutions, licensed research facilities, or organizations with active IRB/IACUC oversight.
Purchaser Restrictions
- Purchaser must be a qualified researcher at an accredited institution or licensed research facility
- This product may not be sold or redistributed to individual consumers, wellness clinics, health food stores, or retail establishments
- Not intended for human or animal consumption, diagnostic use, or therapeutic application
- Institutional affiliation and research purpose will be verified prior to order fulfillment
Distribution is limited to qualified research use in compliance with applicable federal and state law. These products bear the "For Research Use Only" designation per FDA labeling requirements (minimum 10 pt. font). Ref: 21 U.S.C. § 505; FD&C Act § 201(p) (unapproved new drug definition).
| Risk Tier | YELLOW |
| Category | Growth Hormone Axis |
| Subcategory | GH Secretagogue (GHRH Analog) |
| Pharmacological Class | Peptide Hormone Analog |
| Subclass | Growth Hormone-Releasing Hormone (GHRH) Analog |
| Molecular Type | Synthetic Peptide (29 amino acids — the first 29 of the 44-AA endogenous GHRH) |
| Origin | Synthetic analog of endogenous GHRH (somatoliberin) |
| Regulatory Status | FDA-approved as Geref Diagnostic (sermorelin acetate for injection) for diagnostic evaluation of pituitary GH secretion capacity. Previously marketed as Geref (therapeutic) but withdrawn from the US market in 2008 for commercial reasons, not safety. |
| Route of Administration | Subcutaneous injection |
| Reconstitution | Lyophilized powder; reconstitute with bacteriostatic water |
| Storage | Refrigerate (2-8°C) before and after reconstitution |
Chemical Properties
| Molecular Formula | C99H159N37O23 |
| Molecular Weight | 2235.6 g/mol |
| Exact Mass | 2235.2443093 Da |
| InChI Key | ZJCXKXFAOCLSRV-RYLVIXIQSA-N |
| Synonyms |
|
| PubChem | View full record |
Source: NCBI PubChem — public domain data
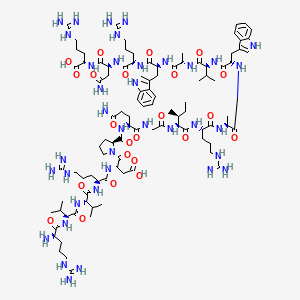
2D structure diagram from NCBI PubChem. This is the actual molecular structure of Sermorelin.
Description
Sermorelin is a synthetic peptide corresponding to the first 29 amino acids of the naturally occurring 44-amino-acid growth hormone-releasing hormone (GHRH), also known as somatoliberin or growth hormone-releasing factor (GRF). Despite being a truncated version, these 29 residues contain the full biological activity of the native molecule — the C-terminal 15 amino acids of endogenous GHRH are not required for receptor binding or signal transduction at the GHRH receptor (GHRH-R) on anterior pituitary somatotroph cells.
The mechanism of action is straightforward and physiological: sermorelin binds to the GHRH receptor on somatotroph cells in the anterior pituitary gland, activating a Gs-protein/adenylyl cyclase/cAMP signaling cascade that stimulates both the synthesis and pulsatile release of endogenous growth hormone (GH). Critically, this process is still subject to normal negative feedback control via somatostatin (growth hormone-inhibiting hormone, GHIH) and IGF-1 — meaning sermorelin does not override the body's regulatory mechanisms. This feedback-preserved mechanism is a key clinical advantage over exogenous GH administration, which bypasses pituitary regulation entirely. The resulting GH release follows a more physiological pulsatile pattern. Sermorelin is considered less potent than CJC-1295 on a per-milligram basis due to its shorter half-life and the lack of modifications that protect it from dipeptidyl peptidase IV (DPP-IV) cleavage.
Clinical Context
Sermorelin holds a unique position as the only GHRH analog to have received FDA approval (as Geref), even though its therapeutic marketing was discontinued for commercial reasons in 2008. This former FDA-approved status provides a stronger regulatory and safety foundation than most other GH secretagogues. In clinical practice and research, sermorelin is often used as a gentler, more physiological alternative to direct GH replacement — particularly in patients with age-related GH decline (somatopause) where complete pituitary function is preserved. It requires a functional pituitary gland to work, which makes it both a diagnostic tool (testing pituitary reserve) and a limitation (it will not work in patients with pituitary damage or failure).
- Requires a functional anterior pituitary gland — will not produce GH release in patients with pituitary insufficiency or destruction
- Former FDA-approved status (Geref) provides extensive human safety data
- The feedback-preserved mechanism means risk of GH excess is significantly lower than with exogenous GH
- Most common adverse effects: injection site reactions, flushing, headache, dizziness
- Evening dosing (before sleep) is preferred to align with the natural nocturnal GH pulse
- DPP-IV rapidly cleaves native sermorelin, contributing to a relatively short half-life (~10-20 minutes); this is why modified analogs like CJC-1295 were developed
- Two SKUs are available: YPB.211 (Premier PLDS, $57.77) and YPB.112 (Standard sourcing, $72.00) — both are 10mg vials
Published Research
Published Research & Clinical Data
Peer-reviewed studies and clinical trial data related to Sermorelin
1 from PubChem
All research below is conducted by independent institutions. MedTech Research Group provides these references for informational purposes only.
Lee-Huang S, Maiorov V, Huang PL, Ng A, Lee HC, et al.. Biochemistry, 2005.PMID: 15779891
18 Registered Clinical Trials
Research data sourced from ClinicalTrials.gov. Public domain (U.S. National Library of Medicine).
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
18
Total Trials
0
Recruiting
0
Active
15
Completed
Sponsor: Theratechnologies · Completed: 2008-10
Sponsor: Massachusetts General Hospital · Completed: 2012-11
Sponsor: Massachusetts General Hospital · Completed: 2010-04
Sponsor: Versartis Inc. · Completed: 2012-07
Sponsor: Theratechnologies · Completed: 2008-07
Research Library — 132 Papers
Research data sourced from OpenAlex. CC0 public domain. Articles are the work of their respective authors.
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
G Protein-Coupled Receptors as Targets for Approved Drugs: How Many Targets and How Many Drugs?
Krishna Sriram, Paul A. Insel · Molecular Pharmacology
Research by Krishna Sriram et al., published in Molecular Pharmacology. Not conducted by MedTech Research Group.
An Official American Thoracic Society/European Respiratory Society Statement: Update on Limb Muscle Dysfunction in Chronic Obstructive Pulmonary Disease
François Maltais, Marc Decramer, Richard Casaburi, et al. · American Journal of Respiratory and Critical Care Medicine
Research by François Maltais et al., published in American Journal of Respiratory and Critical Care Medicine. Not conducted by MedTech Research Group.
G protein-coupled receptors: structure- and function-based drug discovery
Dehua Yang, Qingtong Zhou, Viktorija Labroska, et al. · Signal Transduction and Targeted Therapy
Research by Dehua Yang et al., published in Signal Transduction and Targeted Therapy. Not conducted by MedTech Research Group.
THE CONCISE GUIDE TO PHARMACOLOGY 2017/18: G protein‐coupled receptors
S P H Alexander, Arthur Christopoulos, Anthony P. Davenport, et al. · British Journal of Pharmacology
Research by S P H Alexander et al., published in British Journal of Pharmacology. Not conducted by MedTech Research Group.
Advances in therapeutic peptides targeting G protein-coupled receptors
Anthony P. Davenport, Conor C. G. Scully, Chris de Graaf, et al. · Nature Reviews Drug Discovery
Research by Anthony P. Davenport et al., published in Nature Reviews Drug Discovery. Not conducted by MedTech Research Group.
Mas-Related G Protein-Coupled Receptor-X2 (MRGPRX2) in Drug Hypersensitivity Reactions
Grzegorz Porębski, Kamila Kwiecień, Magdalena Pawica, et al. · Frontiers in Immunology
Research by Grzegorz Porębski et al., published in Frontiers in Immunology. Not conducted by MedTech Research Group.
Growth hormone enhances thymic function in HIV-1–infected adults
Laura A. Napolitano, Diane Schmidt, Michael B. Gotway, et al. · Journal of Clinical Investigation
Research by Laura A. Napolitano et al., published in Journal of Clinical Investigation. Not conducted by MedTech Research Group.
Growth hormone for in vitro fertilization
James MN Duffy, Gaity Ahmad, Lamiya Mohiyiddeen, et al. · Cochrane Database of Systematic Reviews
Research by James MN Duffy et al., published in Cochrane Database of Systematic Reviews. Not conducted by MedTech Research Group.
The Effects of Central Adiposity on Growth Hormone (GH) Response to GH-Releasing Hormone-Arginine Stimulation Testing in Men
Hideo Makimura, Takara L. Stanley, David Mun, et al. · The Journal of Clinical Endocrinology & Metabolism
Research by Hideo Makimura et al., published in The Journal of Clinical Endocrinology & Metabolism. Not conducted by MedTech Research Group.
Growth Hormone Doping in Sports: A Critical Review of Use and Detection Strategies
Gerhard Baumann · Endocrine Reviews
Research by Gerhard Baumann, published in Endocrine Reviews. Not conducted by MedTech Research Group.