SS-31 (Elamipretide)
Research Hub — Aggregated Studies
MedTech Research Group aggregates published research from peer-reviewed journals, clinical trials, and academic institutions. We do not conduct original research. All studies cited below are the work of their respective authors and institutions. Sources are linked for verification.
This product is designated FOR RESEARCH USE ONLY (RUO). These compounds have not been approved or cleared under 21 U.S.C. § 505 and have not been evaluated by the FDA for safety, efficacy, or labeling for clinical, diagnostic, or therapeutic use in humans or animals.
MedTech Research Group will only fulfill orders to qualified researchers affiliated with accredited academic institutions, licensed research facilities, or organizations with active IRB/IACUC oversight.
Purchaser Restrictions
- Purchaser must be a qualified researcher at an accredited institution or licensed research facility
- This product may not be sold or redistributed to individual consumers, wellness clinics, health food stores, or retail establishments
- Not intended for human or animal consumption, diagnostic use, or therapeutic application
- Institutional affiliation and research purpose will be verified prior to order fulfillment
Distribution is limited to qualified research use in compliance with applicable federal and state law. These products bear the "For Research Use Only" designation per FDA labeling requirements (minimum 10 pt. font). Ref: 21 U.S.C. § 505; FD&C Act § 201(p) (unapproved new drug definition).
| Risk Tier | YELLOW |
| Category | Mitochondrial |
| Subcategory | Mitochondrial Inner Membrane Targeting |
| Pharmacological Class | Peptide |
| Subclass | Szeto-Schiller Peptide / Cardiolipin-Targeting Mitochondrial Therapeutic |
| Molecular Type | Synthetic Tetrapeptide (D-Arg-Dmt-Lys-Phe-NH2, where Dmt = 2',6'-dimethyltyrosine) |
| Origin | Synthetic — designed by Dr. Hazel Szeto and Dr. Peter Schiller (hence 'SS' designation) |
| Regulatory Status | Investigational. FDA Fast Track Designation for Barth Syndrome. Phase 2/3 trials conducted by Stealth BioTherapeutics (elamipretide) for multiple mitochondrial indications. Not FDA-approved. |
| Route of Administration | Subcutaneous injection |
| Reconstitution | Lyophilized powder; reconstitute with bacteriostatic water |
| Storage | Refrigerate (2-8°C) |
Chemical Properties
| Molecular Formula | C32H49N9O5 |
| Molecular Weight | 639.8 g/mol |
| Exact Mass | 639.38566570 Da |
| InChI Key | SFVLTCAESLKEHH-WKAQUBQDSA-N |
| Synonyms |
|
| PubChem | View full record |
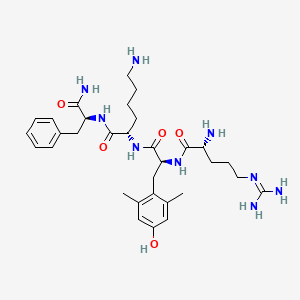
Source: NCBI PubChem — public domain data
2D structure diagram from NCBI PubChem. This is the actual molecular structure of SS-31 (Elamipretide).
Description
SS-31 (elamipretide, also known as Bendavia and MTP-131) is a mitochondria-targeted tetrapeptide that represents a novel class of therapeutics designed to restore mitochondrial function at the level of the inner mitochondrial membrane. Unlike traditional antioxidants that attempt to scavenge reactive oxygen species (ROS) after they are produced, SS-31 targets the source of mitochondrial dysfunction by selectively binding to cardiolipin — a unique phospholipid found exclusively in the inner mitochondrial membrane.
Cardiolipin is essential for the structural organization and function of the electron transport chain (ETC) complexes. It interacts with Complexes I, III, IV, and the ATP synthase (Complex V), and it is critical for the formation of respiratory supercomplexes (respirasomes) that optimize electron transfer efficiency. When cardiolipin becomes oxidized — as occurs with aging, ischemia, and mitochondrial disease — the ETC complexes destabilize, electron transfer becomes inefficient, ROS production increases (creating a vicious cycle of further cardiolipin oxidation), cytochrome c detaches from the inner membrane (triggering apoptosis), and ATP production declines. SS-31 binds selectively to cardiolipin through electrostatic interactions (the alternating aromatic-cationic motif in its structure: Arg+-Dmt-Lys+-Phe), stabilizing cardiolipin's interaction with ETC complexes, preventing cytochrome c peroxidase activity, optimizing electron transfer, and reducing ROS generation at the source. The net effect is restoration of mitochondrial bioenergetics — improved ATP production, reduced oxidative stress, and protection against apoptotic signaling.
SS-31 has a unique cell-penetrating property: it concentrates in mitochondria at 1000-5000x the extracellular concentration within minutes, driven by the mitochondrial membrane potential. This concentration effect means that very low systemic doses can achieve therapeutically relevant mitochondrial concentrations.
Clinical Context
SS-31/elamipretide has been studied in multiple clinical trials by Stealth BioTherapeutics for mitochondrial myopathy (MMPOWER trials), Barth Syndrome (TAZPOWER), heart failure (PROGRESS-HF), age-related macular degeneration, and renal diseases. The FDA granted Fast Track Designation for Barth Syndrome, a rare X-linked cardioskeletal mitochondrial disease caused by mutations in the tafazzin gene (which is required for cardiolipin remodeling). Clinical trial results have been mixed — some endpoints showed improvement while others did not reach statistical significance — but the safety profile has been consistently favorable. The mitochondrial medicine field remains highly active, and SS-31 continues to be one of the most pharmacologically advanced mitochondria-targeted agents.
- Targets the inner mitochondrial membrane (cardiolipin), not ROS scavenging — mechanistically distinct from traditional antioxidants
- Concentrates 1000-5000x in mitochondria — very low systemic doses are effective
- FDA Fast Track for Barth Syndrome indicates regulatory interest in the mechanism
- Well-tolerated in clinical trials; most common adverse effect is injection site reactions
- Two vial sizes available: 10mg ($33.59) for initial research and 50mg ($124.11) for extended protocols
- Not a general antioxidant — specifically targets mitochondrial bioenergetics
- May be particularly relevant for conditions with established mitochondrial dysfunction (aging, heart failure, neurodegenerative disease, metabolic syndrome)
- The alternating aromatic-cationic motif (Arg-Dmt-Lys-Phe) is the pharmacophore — modifications that disrupt this pattern eliminate mitochondrial targeting
Published Research
Published Research & Clinical Data
Peer-reviewed studies and clinical trial data related to SS-31 (Elamipretide)
12 from PubChem
All research below is conducted by independent institutions. MedTech Research Group provides these references for informational purposes only.
Han Z, Varadharaj S, Giedt RJ, Zweier JL, Szeto HH, et al.. The Journal of pharmacology and experimental therapeutics, 2009.PMID: 19131585
Reddy TP, Manczak M, Calkins MJ, Mao P, Reddy AP, et al.. International journal of environmental research and public health, 2011.PMID: 21318024
Birk AV, Liu S, Soong Y, Mills W, Singh P, et al.. Journal of the American Society of Nephrology : JASN, 2013.PMID: 23813215
Szeto HH. British journal of pharmacology, 2014.PMID: 24117165
Liu X, Wu J, Wang J, Feng X, Wu H, et al.. Human & experimental toxicology, 2020.PMID: 31884831
Arsenic Directs Stem Cell Fate by Imparting Notch Signaling Into the Extracellular Matrix Niche.
Anguiano T, Sahu A, Qian B, Tang WY, Ambrosio F, et al.. Toxicological sciences : an official journal of the Society of Toxicology, 2020.PMID: 32647881
Zhang H, Ran M, Jiang L, Sun X, Qiu T, et al.. Ecotoxicology and environmental safety, 2023.PMID: 37979351
Li M, Kong D, Meng L, Wang Z, Bai Z, et al.. RSC advances, 2024.PMID: 39301232
Elamipretide: A Review of Its Structure, Mechanism of Action, and Therapeutic Potential.
Tung C, Varzideh F, Farroni E, Mone P, Kansakar U, et al.. International journal of molecular sciences, 2025.PMID: 39940712
Dong LL, Jia XQ, Xie HB, Zhu LF, Zhou PC, et al.. Toxicology and applied pharmacology, 2025.PMID: 40669774
Shirley M. Drugs, 2026.PMID: 41335372
Haefely W, Ruch-Monachon MA, Jalfre M, Schaffner R. Arzneimittel-Forschung, 1976.PMID: 9104
28 Registered Clinical Trials
Research data sourced from ClinicalTrials.gov. Public domain (U.S. National Library of Medicine).
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
28
Total Trials
1
Recruiting
1
Active
20
Completed
Sponsor: Stealth BioTherapeutics Inc. · Completed: 2017-06-02
Sponsor: Stealth BioTherapeutics Inc. · Completed: 2020-02-10
Sponsor: Stealth BioTherapeutics Inc. · Completed: 2021-10-11
Sponsor: Stealth BioTherapeutics Inc. · Completed: 2016-07-06
Sponsor: Stealth BioTherapeutics Inc. · Completed: 2024-12-04
Research Library — 725 Papers
Research data sourced from OpenAlex. CC0 public domain. Articles are the work of their respective authors.
MedTech Research Group provides these references for informational purposes. We do not conduct original research. All studies are the work of their respective authors and institutions.
Mitochondrial energetics in the kidney
Pallavi Bhargava, Rick G. Schnellmann · Nature Reviews Nephrology
Research by Pallavi Bhargava et al., published in Nature Reviews Nephrology. Not conducted by MedTech Research Group.
Mitochondrial dysfunction and oxidative stress in heart disease
Jessica N. Peoples, Anita Saraf, Nasab Ghazal, et al. · Experimental & Molecular Medicine
Research by Jessica N. Peoples et al., published in Experimental & Molecular Medicine. Not conducted by MedTech Research Group.
Mitochondrial function as a therapeutic target in heart failure
David A. Brown, Justin B. Perry, Mitchell E. Allen, et al. · Nature Reviews Cardiology
Research by David A. Brown et al., published in Nature Reviews Cardiology. Not conducted by MedTech Research Group.
Oxidative stress in the aging substantia nigra and the etiology of Parkinson's disease
Benjamin G. Trist, Dominic J. Hare, Kay L. Double · Aging Cell
Research by Benjamin G. Trist et al., published in Aging Cell. Not conducted by MedTech Research Group.
Mitochondrial quality control in kidney injury and repair
Chengyuan Tang, Juan Cai, Xiao-Ming Yin, et al. · Nature Reviews Nephrology
Research by Chengyuan Tang et al., published in Nature Reviews Nephrology. Not conducted by MedTech Research Group.
Role of Cardiolipin in Mitochondrial Function and Dynamics in Health and Disease: Molecular and Pharmacological Aspects
Giuseppe Paradies, Valeria Paradies, Francesca Ruggiero, et al. · Cells
Research by Giuseppe Paradies et al., published in Cells. Not conducted by MedTech Research Group.
Protein posttranslational modifications in health and diseases: Functions, regulatory mechanisms, and therapeutic implications
Qian Zhong, Xina Xiao, Yijie Qiu, et al. · MedComm
Research by Qian Zhong et al., published in MedComm. Not conducted by MedTech Research Group.
Repairing Mitochondrial Dysfunction in Disease
Vincenzo Sorrentino, Keir J. Menzies, Johan Auwerx · The Annual Review of Pharmacology and Toxicology
Research by Vincenzo Sorrentino et al., published in The Annual Review of Pharmacology and Toxicology. Not conducted by MedTech Research Group.
Ischemia and Reperfusion Injury in Kidney Transplantation: Relevant Mechanisms in Injury and Repair
Gertrude J. Nieuwenhuijs‐Moeke, Søren Erik Pischke, Stefan P. Berger, et al. · Journal of Clinical Medicine
Research by Gertrude J. Nieuwenhuijs‐Moeke et al., published in Journal of Clinical Medicine. Not conducted by MedTech Research Group.
Mitochondrial medicine in the omics era
Joyeeta Rahman, Shamima Rahman · The Lancet
Research by Joyeeta Rahman et al., published in The Lancet. Not conducted by MedTech Research Group.